1) Ethoxyethane, C2H5OC2H5, is commonly used as a solvent in the purification of compounds. The boiling point of C2H5OC2H5 is 36 °C. The safety data sheet for C2H5OC2H5 states: ‘Extremely flammable. Keep away from sources of ignition.’ During the purification process, a compound is dissolved in C2H5OC2H5 by heating it for an extended period of time. This is done using glassware that is open to the atmosphere. This step in the purification process should be carried out using a
A. water bath in a fume cupboard.
B. water bath on a laboratory bench.
C. Bunsen burner in a fume cupboard.
D. Bunsen burner on a laboratory bench.
Solution

2) At the molecular level, Protein P is shaped like a coil. When a solution of Protein P is mixed with citric acid, solid lumps form. The change in the structure of Protein P is due to
A. hydrolysis.
B. denaturation.
C. polymerisation.
D. the formation of peptide bonds.
Solution
3) Which one of the following fatty acids is an omega-3 fatty acid?
A. arachidonic
B. palmitoleic
C. linolenic
D. linoleic
Solution

4) The following table contains the percentage composition by mass of the nutritional value of some common foods.
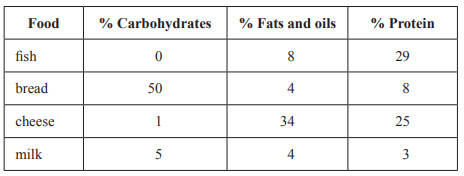 .
.
Which one of the following servings has the highest energy content?
A. 100 g of fish
B. 80 g of bread
C. 40 g of cheese
D. 258 g (250 mL) of milk
Solution

5) Which one of the following molecules contains a chiral carbon?
A. CH2CHCH2CH3
B. CH2FCH2CH2Cl
C. CH3CHOHCH2CH3
D. CH3CH2CFClCH2CH3
Solution

6) A student wants to use a physical property to distinguish between two alcohols, octan-1-ol and propan-1-ol. Both alcohols are colourless liquids at standard laboratory conditions (SLC). The student should use
A. density because propan-1-ol has a much higher density than octan-1-ol.
B. boiling point because octan-1-ol has a higher boiling point than propan-1-ol.
C. electrical conductivity because octan-1-ol has a higher conductivity than propan-1-ol.
D. spectroscopy because it is not possible to distinguish between the alcohols using their physical properties.
Solution

7) Which one of the following is a dipeptide made from α-amino acids?
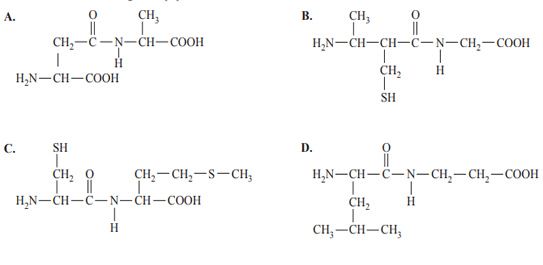
Solution

8) Organic compounds are numerous and diverse due to the nature of the carbon atom. There are international conventions for the naming and representation of organic compounds.
a. i. Draw the structural formula of 2-methyl-propan-2-ol.
Solution
ii. Give the molecular formula of but-2-yne.
Solution

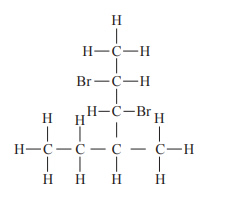
b. Give the IUPAC name of the compound that has the structural formula shown above.
Solution

c. The following diagram represents a reaction pathway for the synthesis of Compound P.
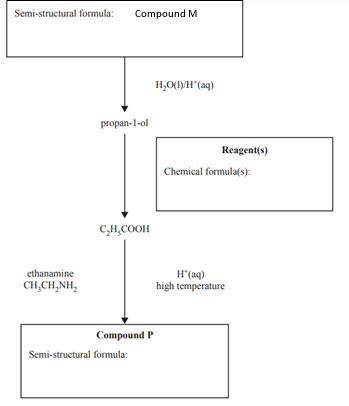
i. Identify the starting substance, Compound M, by writing its semi-structural formula in the box provided.
ii. Identify the reagent(s) needed to convert propan-1-ol to propanoic acid,
C2H5COOH, by writing the chemical formula(s) of the reagent(s) in the box provided.
iii. When C2H5COOH, is mixed with ethanamine, CH3CH2NH2, in an acidified high-temperature environment, Compound P is formed. Write the semi-structural formula of Compound P in the box provided
Solution

9) There are four optical isomers of ascorbic acid. Only one of these, L-ascorbic acid (vitamin C), is active in the human body. Vitamin C is essential in the human diet and has many functions. One of its functions is as a coenzyme in the production of the protein collagen. During the synthesis of collagen, vitamin C acts as an electron donor. Collagen is important in the body. A lack of collagen results in a condition called scurvy.
a. The second most common amino acid in collagen is proline. Draw the zwitterion of proline.
Solution
b. Describe the role of vitamin C as a coenzyme in collagen synthesis. Use the physical and chemical interactions of vitamin C with the enzyme to explain how the enzyme is able to catalyse the production of collagen
Solution

C. i. What is meant by the term ‘optical isomer’?
Solution
ii. Explain why L-ascorbic acid, as vitamin C, is active as a coenzyme in the human body while the other optical isomers are not.
Solution

d. Vitamin C can also act as an antioxidant and is often added to food. Explain why foods that contain a high proportion of unsaturated fats would require more vitamin C to preserve them than foods that contain a high proportion of saturated fats.
Solution
